What Is The TCT?
Every fall since 1988, audiences of interventional cardiologists
have gathered in Washington, DC for the annual TCT (Transcatheter
Cardiovascular Therapeutics) Symposium. This year's meeting, attended
by an estimated 12,000 physicians, spanned 5 days and included
scores of presentations, dozens of live case demonstrations broadcast
from around the world, the announcement of a dozen late breaking
clinical trials, exhibits from over 300 device and other manufacturers,
and special evening symposia -- all presented by an international
faculty of hundreds of interventional cardiologists and their radiological
and surgical colleagues. The event receives substantial support
from the medical industry and serves as a showcase for the latest
developments in interventional vascular therapy. |
|
 TCT
symposium is held annually at the Washington Convention
Center
TCT
symposium is held annually at the Washington Convention
Center |
Quick Summary
While no revolutionary announcements were made during the meeting, the
information and presentations did clarify certain directions in therapy,
directions that will have significant benefit to patients:
- Positive results for drug-eluting
stents (a.k.a coated stents or medicated
stents) continued to be reported -- the benefits
of these devices are proving to be long-lasting,
or what interventionalists call "durable",
and the devices are expanding in application -- good
results were shown for complex coronary cases, and
in patients with diabetes, patient populations previously
not considered optimal for stenting;
- Interventional catheter-based
therapy is expanding to treat vascular disease outside
of the heart -- very positive reports on carotid
stenting (opening up the arteries in the neck that
feed the brain) with results comparable or better
than the most commonly-used "open" surgical
procedure of carotid endarterectomy;
- Several novel devices for specialized
situations, such as the SafeCross® infra-red/radio
frequency catheter that allows the penetration of
total occlusions in the coronary arteries;
- Results of several studies that
put to rest certain therapies/devices, such as brachytherapy,
or intracoronary radiation, that have not wound up
being as effective as originally thought.
|
 Taxus
Stent
Taxus
Stent
(courtesy Boston Scientific) |
|
DES
(Drug-Eluting Stents)
Clearly the most important recent advance
in the field of coronary angioplasty has been the drug-eluting
stent. Currently two such stents are approved for use
in the U.S.: the Cypher™ from Cordis/Johnson & Johnson
and the Taxus™ from Boston Scientific. A number
of studies and clinical trial results reported continued
efficacy for these devices which reduce the restenosis
or reclosing of the coronary artery after stent placement
from 20-30% down to single digits. While the differences
in these studies are closely watched by investors,
marketing departments and others who have vested interests
in the specific manufacturing companies, these differences
pale when one compares the overall performance of all
drug-eluting stents to the older bare metal stents.
|
DES:
Multivessel Disease
Patrick Serruys, MD, of the ThoraxCenter in Rotterdam presented the results
of ARTS II (Arterial Revascularization Therapies II). The original ARTS
I study, done in 1997 before drug-eluting stents were available, compared
bypass surgery to bare metal stents. ARTS II added in a patient component
that received the Cypher stent. All patients were treated for multiple
vessel coronary disease, a situation in which bypass surgery is often
recommended over angioplasty. However, the MACE (Major Adverse Coronary
Event) rate after 6-months showed that only 7% of the Cypher group suffered
an event, as compared to 9% of the surgical group and 20% of those who
had bare metal stents. A MACE is defined as heart attack, stroke, death,
or repeat surgery/angioplasty.
DES:
Diabetic Patients
Dr. Keith D. Dawkins
of Southampton General Hospital in England began his
evening presentation with a frightening statistic, showing
a five-fold increase in diabetes mellitus from 1985 to
2000, increasing to a ten-fold increase predicted over
the next twenty years. He called diabetes nothing less
than a "pandemic". And diabetic patients are
at significantly greater risk of developing coronary
artery disease. The conundrum has always been, however,
that interventional therapies, such as angioplasty, have
been less successful for diabetic patients than for non-diabetics.
Restenosis rates (30-40%) and other complications have
caused many physicians to recommend open heart surgery
instead. |
|
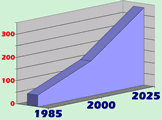 Diabetics
Worldwide in Millions
Diabetics
Worldwide in Millions
-- International Diabetes Federation |
However, drug-eluting stents may be changing
that scenario. Dr. Manel Sabaté of Hospital Clinico
San Carlos, Madrid, Spain presented the DIABETES trial which
compared the Cypher to a bare metal stent in diabetic patients.
Restenosis rates were only 7.7% for the coated Cypher stent,
while the bare metal stent reclosed an expected 33% of the
time. Data from the e-Cypher registry also confirmed good
results in diabetic patients. Dr. Giulio Guagliumi, of the
Ospedali Riuniti de Bergamo in Italy, stated that the TLR
(target lesion revascularization) for diabetics was only
1.67% higher than that for non-diabetic patients. As for
the Taxus coated stent, Gregg Stone, MD, of Columbia University
Medical Center in New York presented two-year followup for
the Taxus IV study, showing diabetic patients with a TLR
rate of 8%. The bottom line here is that drug-eluting stents
seem to have great benefits for diabetic patients. Certainly
more studies need to be done and there are other measures
which need to be considered, but it seems that the drugs
on these stents interrupt the response that causes restenosis
in diabetics.

"Stents have narrowed the gap...Drug-eluting stents, now with more data
coming, should erase the differences."
-- Martin Leon, MD |
|
DES
vs. Surgery
Additionally, the Taxus studies showed that better than delaying
the onset of restenosis, the drug-eluting stents seemed to be preventing
it -- the benefits of the Taxus stents actually increased over the
two-year study period. All this new research prompted Dr. Martin
Leon, organizer of the TCT, to state, "Stents have narrowed
the gap in mortality benefit in CABG [bypass surgery] vs. PCI [angioplasty].
Drug-eluting stents, now with more data coming, should erase the
differences.” And in an evening symposium, co-organizer Dr.
Gregg Stone speculated whether, if results from these future studies
were comparable to the early indications, at some point in the not-too-distant
future, these minimally invasive procedures might end up replacing
most bypass surgery. |
Carotids
and Beyond
On the heels of the FDA approval of Guidant's RX ACCULINK™ Carotid
Stent System and RX ACCUNET™ Embolic Protection System last month,
two studies on carotid angioplasty/stenting were presented. In this procedure,
the artery in the neck that leads to the brain, is opened in much the
same way that a coronary angioplasty is performed. Today the carotid
is usually opened surgically in an procedure called carotid endartarectomy.
But in many centers, stenting is now being performed, especially in compromised
patients who may not do well in surgery. The clinical trials presented
were named CABERNET and the MAVERIC II (suffice it to say that these
acronyms refer to the devices used). The CABERNET trial was a combination
of the NextStent™ by EndoTex and the FilterWire™ EX/EZ made
by Boston Scientific. The MAVERIC trial utilized the Exponent stent and
the GuardWire™ balloon occlusion system, both made by Medtronic.
In each system, a stent is expanded in the carotid artery while a filter
or balloon prevents any debris loosened by the angioplasty from travelling
to the brain. The results exceeded expectations and showed the minimally
invasive carotid stenting to be equal or better than conventional surgery.
There are still many questions -- the sample size of the studies was
relatively small -- but the results are superior to similar studies done
over the past few years, implying that doctors are getting better at
performing the procedure, AND that the devices themselves are being improved
by the manufacturers.
Angioplasty/stenting in the kidneys and
the legs were also discussed -- as was an even newer application
of endovascular therapy, the treatment of AAA (Abdominal
Aortic Aneurysm) with stent grafts. The move in medicine
today is clearly away from conventional open surgery and
more and more towards endovascular or interventional solutions
(angioplasty and stenting). For more on this evolution/revolution,
view our documentary (now on DVD) "Vascular Pioneers:
Evolution of A Specialty" at VascularTherapy.Org.
Intracoronary
Radiation Studies Show No Benefit
As a check to over-enthusiasm about new technologies that have not
yet been studied long-term, a sobering study was presented. The five-year
follow-up from the Gamma-1 study of the Cordis CHECKMATE™ Gamma
Radiation System (brachytherapy) showed disappointing results for
reducing in-stent restenosis (stents that have been clogged). While
the results at two-years looked somewhat positive, over five-years
the curves of the patients treated with radiation and placebo converged,
so that there was no perceived benefit. The results were not a great
surprise to many cardiologists and the use of intravascular radiation
has been dwindling (Guidant discontinued its GALILEO® Intravascular
Radiotherapy System back in April 2004). |
|
 radiation
catheter
radiation
catheter
(courtesy Cordis) |
Dr. Leon did not think the same problem
will be found in drug-eluting stents. He stated to theHeart.org's
HeartWire, "We view vascular brachytherapy as if
you're basically exploding a bomb in the coronary arteries...You
eternally alter the biology of the vessel wall, you affect
endothelialization, you affect platelet absorption...and
you affect the healing response...We view DES not as a bomb,
but more as a laser beam...We think that the effects are
shorter, more specific, and we're hopeful that the effects
will be much more durable." He concluded that in his
hospital, "We have essentially ceased using vascular
brachytherapy."
For the latest news about developments
in the field, go to our Drug-Eluting
Stent NewsCenter. We welcome your
comments on this article.
Source: Angioplasty.org, October 11, 2004
CONTACT: Burt
Cohen, Producer of Angioplasty.org
|