|
Medtronic Receives
FDA Approval of Talent Captivia® System for Endovascular Treatment
of Thoracic Aortic Aneurysms
New Tip Capture Feature
Enables Controlled Deployment and Precise Placement of Stent Graft
During Minimally Invasive Procedure to Repair Bodys Main Artery
|
 |
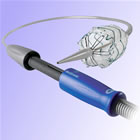
Medtronic
Captiva
Delivery System |
|
November 1, 2010 --
Minneapolis --
Advancing the minimally invasive treatment of aortic aneurysms,
Medtronic, Inc. (NYSE: MDT), today announced approval by the
U.S. Food and Drug Administration (FDA) of the Talent Thoracic
Stent Graft with Captivia® Delivery System, which features
a tip capture mechanism for controlled deployment and precise
placement
of the implantable medical device. Now available for clinical practice
in the United States, the Talent Captivia System is used in
the endovascular repair of
thoracic aortic aneurysms (TAA), dangerous bulges in the bodys
main artery (near the heart) that can rupture with fatal consequences
if left untreated.
|
While an estimated 60,000 people in the United
States have a TAA, only about half are ever diagnosed due to lack
of symptoms.
During thoracic endovascular
aortic repair (TEVAR), the Talent Captivia System is inserted
into the femoral artery in the patients groin and moved up through
blood vessels to the aorta. With the device at the site
of the aneurysm, the physician expands the stent graft within
the aorta,
creating a new path for blood flow that reduces pressure on the
bulge and the risk of rupture.
The Captivia Delivery Systems
tip capture mechanism is designed to provide excellent control
of the stent graft during deployment to ensure that blood flow
isnt occluded into the nearby arteries, said Dr. Edward Y.
Woo, M.D., vice-chief and program director of vascular surgery
and endovascular therapy for the University of Pennsylvania Hospital
System. This improvement to the delivery system also increases
my confidence in the devices deployment accuracy.
Medtronic now offers the Talent Thoracic Stent Graft in longer
lengths (up to 215 mm in total length) to accommodate the aortic
anatomies of more patients. In addition to tip capture, the
Captivia Delivery System features a hydrophilic coating to
ease insertion into the femoral artery and navigation through
the iliac arteries (around the pelvis) en route to the aorta. |
|
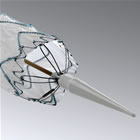
Medtronic
Captiva Delivery System (Tip Detail) |
In partnership with physicians around the world, Medtronic stent grafts have been used to treat more than 180,000 patients with aortic disease or injury more than all other manufacturers combined, said Tony Semedo, vice president and general manager of Medtronics Endovascular Innovations business. FDA
approval of the Talent Captivia System builds on this legacy
of leadership in the field of endovascular aortic repair. The Talent Captivia System is indicated specifically for the endovascular repair of fusiform aneurysms and saccular aneurysms/penetrating ulcers of the descending thoracic aorta in patients having appropriate anatomy. More information about Medtronic stent grafts, including the Talent Captivia System, is available online at www.medtronicendovascular.com. Medtronic is committed to advancing the treatment of coronary, peripheral, aortic and structural heart disease through collaboration with leading clinicians, researchers and scientists worldwide.
About Medtronic
Medtronic, Inc. (www.medtronic.com),
headquartered in Minneapolis, is the global leader in medical technology alleviating
pain, restoring health and extending life for millions of people
around the world. Any forward-looking
statements are subject to risks and uncertainties such as those
described
in Medtronics periodic reports on file with the Securities and
Exchange Commission. Actual results may differ materially from
anticipated results.
Source: Medtronic,
Inc.
|


