November 19, 2010 --
Minneapolis and New York -- An implantable medical
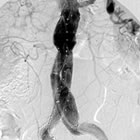
device used in the minimally invasive treatment of abdominal
aortic aneurysms, the Endurant® Stent
Graft System from Medtronic, Inc. (NYSE: MDT), delivered strong
results through one year of patient follow-up in the companys
U.S. pivotal study, according to clinical data presented at VEITHsymposium.
Approved by the U.S. Food and Drug Administration (FDA) under
an investigational device exemption (IDE), the prospective
study involved
150 patients at 26 U.S. medical centers and met its primary endpoints. |