
|
|
|
|
Medtronic Advances Research on Peripheral Devices, Highlights Upcoming Presentations
EuroPCR Program Includes Endovascular Late-Breakers on IN.PACT Drug-Eluting Balloons and Mo.Ma Ultra Embolic Protection System
Enrollment in IN.PACT SFA I DEB Study Completed;
PIs for IN.PACT
SFA II Study Selected
|
 |
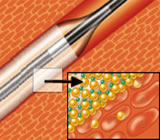
IN.PACT
Drug-Eluting Balloon |
|
May 16, 2011 -- Minneapolis --
A global leader in advancing the treatment of cardiovascular
disease through innovative medical technology, Medtronic, Inc.
(NYSE: MDT) today announced recent milestones in its clinical
program for the IN.PACT drug-eluting balloon (DEB) technology.
The company also announced upcoming data presentations on its
peripheral vascular devices specifically, the IN.PACT DEB
technology for the treatment of atherosclerosis in leg arteries
and the Mo.Ma Ultra proximal cerebral protection device for carotid
artery stenting at the EuroPCR meeting in Paris, May 1720
at the Palais des Congrès. |
IN.PACT DEB Clinical Program Update
Enrollment in the IN.PACT SFA I study was completed on April 29. IN.PACT SFA I is a prospective, multicenter, randomized study comparing the IN.PACT Admiral DEB to standard balloon angioplasty for treatment of de novo and restenotic lesions in the superficial femoral and proximal popliteal arteries.
The study enrolled 150 subjects at 13 sites in Europe. The primary safety endpoint is a composite of 30-day death or target limb major amputation or clinically-driven target vessel revascularization (TVR) within 12 months. The primary efficacy endpoint is primary patency within 12 months. The principal investigator for SFA I is Dr. Gunnar Tepe of the Academic Hospital of Rosenheim in Germany.
The IN.PACT SFA I study forms the basis for the upcoming global IN.PACT SFA II study. SFA II will be led by two principal investigators, interventional cardiologist Dr. John Laird of the UC Davis Medical Center in California and vascular surgeon Dr. Peter Schneider of Kaiser Permanente in Hawaii.
These groundbreaking trials on drug-eluting balloons will help build the evidence base for this promising therapeutic technology, Dr. Tepe said about the latest developments in the IN.PACT clinical program. Drug-eluting
balloons may represent the next-generation therapy for lower extremity
vascular disease as the devices allow interventionalists to treat patients
without placing a permanent implant and maintain future treatment alternatives.
Medtronic is clearly leading this exciting field with its IN.PACT technology
and family of clinical trials.
EuroPCR Endovascular Late-Breakers
Medtronic peripheral devices are the subject of three late-breaking
clinical trials during the EuroPCR Endovascular session titled Whats new in endovascular interventions, that
starts on Tuesday, May 17 at 12:00 CEST in Room Maillot.
- First, at approximately 12:15 CEST, Dr. Alberto
Cremonesi of Villa Maria Cecilia Hospital in Cotignola Ravenna, Italy will
present 30-day results
from the prospective, multicenter DESERVE study: diffusion weighted - magnetic
resonance imaging based evaluation of the effectiveness of endovascular clamping
during carotid artery stenting with the Mo.Ma device. Results from a prospective,
multicentre study. Besides
the typical 30-day neurological assessment, incidence of brain injuries as
detected by dwMRI were investigated in 127 patients who underwent carotid
stenting with use of the Mo.Ma Ultra proximal protection device in combination
with the Cristallo Ideale stent.
- Later, at approximately 12:43 CEST, Dr.
Thilo Tuebler from Herzzentrum Hamburg in Germany will present one-year
results from the MARIS 1,000-patient SFA registry, the largest SFA-stent
registry
ever conducted, now complete with target lesion revascularization and restenosis
data at one year.
- Finally, at approximately 12:57 CEST, Dr. Antonio Micari of Maria Eleonora
Hospital in Palermo, Italy will present 12-month results from the multicenter
DEB SFA IT Registry. This physician-sponsored registry analyzed the performance
of the IN.PACT Admiral DEB in 105 patients with symptoms of claudication
and rest pain due to superficial femoral and popliteal artery disease.
Primary patency results will be presented jointly with meaningful clinical
endpoints,
including walking capacity and quality of life.
In collaboration with leading clinicians, researchers and scientists worldwide, Medtronic offers the broadest range of innovative medical technology for the interventional and surgical treatment of cardiovascular disease and cardiac arrhythmias.
About Medtronic
Medtronic, Inc. (www.medtronic.com), headquartered in Minneapolis, is the global
leader in medical technology alleviating pain, restoring health and extending
life for millions of people around the world.
Any forward-looking statements are subject to risks and
uncertainties such as
those described in Medtronics periodic reports on file with the Securities and
Exchange Commission. Actual results may differ materially from anticipated results.
Source: Medtronic,
Inc.
|
|

