![]()
<< To Homepage >>
<<Archives>>
September-October 2010 Archives:
October 27, 2010 -- 8:55pm EDT To Stent or Not to Stent: That is the Question! Suffice it to say that Dr. Midei does not believe he implanted stents unnecessarily. And his opinion is backed up by two Bills (Dr. William O'Neill of University of Miami and Dr. William Knopf of Piedmont in Atlanta) who were retained by Midei's legal team to review these cases. And who are these cardiologists? Well, Dr. O'Neill often has been quoted on Angioplasty.Org, because he is one of the true pioneers of interventional cardiology, credited with documenting and disseminating the benefits of primary angioplasty as the "gold standard" of treatment for heart attacks (see "Heart Attacks and Angioplasty") -- because of angioplasty and stents, death from heart attacks has been reduced from 25% fifty years ago to 2% today! Dr. Knopf likewise is a widely respected interventionalist who is the COO of the prestigious Piedmont Heart Institute. They have said that they did not see any "unneeded" procedures. Of course, St. Joseph's also retained cardiologists to review the angiograms, among them Dr. Donald Cutlip of Beth Israel Deaconess in Boston, who disagreed with Midei's decision to stent in a number of cases. (Dr. Cutlip is the Executive Director of Clinical Investigation at HCRI, and is widely known for his role in clarifying and setting the current definitions for "stent thrombosis".) So is this a "he said; she said" situation? Or is there some scientific method for determining whether or not a patient needs a stent? One would hope there is, since angioplasty is now 33 years old!! Well, of course, there is -- sort of. I revert to "sort of" because in medicine, we are dealing with human physiology, where there are very few absolutes. But, in the case of the necessity for stents, there actually has been research and clinical trials have been conducted, and the results were quite clear: in January of 2009, the FAME study was published...and it showed that 1/3 of blockages that were judged as "significant" by looking at an angiogram were, in fact, not significant! Using Fractional Flow Reserve (FFR) the actual blood flow through the coronary artery was measured -- and in 1/3 of the cases that looked blocked on the x-ray, they weren't. A bit scary -- to see an angiogram that shows a significant narrowing, and then not stent it? Well the explanation is a bit complicated, but suffice it to say that flow dynamics show that just because a two-dimensional shadow image (the kind produced by an angiogram) implies a narrowing, doesn't mean the blood flow is actually restricted. And FAME's two year results verify this -- of the approximately 500 blockages that looked significant on angiography but were judged not significant via FFR, and were subsequently NOT TREATED, only one went on to cause a myocardial infarction: a very low rate of 0.2%. (It is also the case that a blockage that looks insignificant on an angiogram in fact may be limiting blood-flow and causing ischemia.) Unfortunately in the Maryland Stent Story, FFR was not available at St. Joseph's -- a fact that has also become a point of controversy: the hospital accuses Dr. Midei of not using FFR technology but, according to theheart.org article, Dr. Midei claims:
So the take-away here is that FFR has a number of benefits: one-third less stents, one-third better outcomes -- and should be used everywhere! Yet only 15% or so of cath labs currently have it installed. There are currently two manufacturers of FFR -- Volcano Corporation and St. Jude Medical (as a result of St. Jude's acquisition of Radi Medical Systems in 2008). I wrote about this this past summer in my blog post, "The Un-Stent Wars: Call Out the Fractional Flow Reserves". In any case, one would think that this technology would be more widely used. Perhaps, as a result of controversies like Dr. Midei's, it will be adopted, if only as a way of justifying the placement, or non-placement, of a stent. And, oh yeah, benefiting patients as well! « permalink » « send comment » « back to top »
October 20, 2010 -- 5:00pm EDT Angioplasty Live! The photo above shows Dr. Andreas Gruentzig in his catheterization lab during a live course at Emory Hospital in Atlanta, where he conducted a number of broadcasts in the early 1980's. His purpose was to train cardiologists in this radical new procedure that he had invented in 1977: balloon angioplasty. Gruentzig was adamant that physicians needed to be well-trained and cautious when beginning to do these new types of procedures. Because he knew that unbridled expansion of his technique would result in complications and poor outcomes -- and that would stifle the development of this minimally invasive treatment for coronary artery disease. The years intervening have changed both the purpose and presentation of these live courses considerably. 1978 saw the first of these courses, when Gruentzig, then at University Hospital in Zurich, could not accomodate all the physicians who wanted to learn his technique via visits to his cath lab.
At last month's TCT meeting in Washington, 10,000 attendees watched more than 100 hours of live cases, beamed in from Germany, France and 17 other international sites -- and in high definition on a 150' screen! (photo above from PSAV Presentation Services.) Quite a change in a little over three decades. So the regulatory agencies and professional societies have become concerned that live courses, which have become the cornerstone of many highly-attended cardiology meetings, be done in an ethical, safe and rational manner; that they do not become merely showcases for new equipment, whose manufacturers have contributed to the bottom line of the meeting; that the safety and privacy of the patients involved are protected. These are valid concerns and the joint statement is a good touchstone -- all would be welcomed by Andreas Gruentzig. But the very early angioplasty courses, many of which I had the honor of producing and directing, were a bit different than today. As Dr. Richard Myler, who performed the first angioplasty in the U.S. (simultaneously with Dr. Simon Stertzer) says in the video below:
I created the video below to accompany a discussion of this topic during the 2009 CRT meeting in Washington, as part of the FDA Workshop. It gives a taste of what these early courses were like -- where the patients, instead of having their privacy protected, sat up proudly after the successful procedure and gladly waved with a smile to the audience of cardiologists, a group that would go on to make angioplasty the gold standard for the treatment of heart attacks.
(Disclosures: the creation of the above video was supported by an unrestricted educational grant from Abbott Vascular. Speaking in the video are John E. Abele, co-founder of Boston Scientific, Maria Schlumpf, Gruentzig's assistant, Dr. Spencer B. King, III, Dr. Richard K. Myler, Dr. Gary S. Roubin and Dr. Martin B. Leon. Music by Nell Shaw Cohen.) « permalink » « send comment » « back to top »
October 5, 2010 -- 7:10pm EDT Go With the Flow: Fractional Flow Reserve,
That Is Last November, the American Heart Association (AHA), American College of Cardiology (ACC) and the Society for Cardiac Angiography and Interventions (SCAI) updated their official guidelines, giving Fractional Flow Reserve (FFR) a Class "A" level of evidence. This past August, the European Society of Cardiology updated their official guidelines, boosting FFR to Class 1, level of evidence A -- the highest possible endorsement for this measurement modality. However, only about 15% of cath labs in the U.S. use this technology. So why aren't more using FFR, a functional measurement device which has been proven to reduce healthcare costs? As the FAME study showed, using FFR can cut the use of stents by one-third! Well, it has everything to do with reimbursement and the odd way in which that is applied in the U.S. For example, a cardiologist currently gets a small $99 Medicare payment (CPT® code 93571-26) for performing FFR; it's $78 for an additional vessel (CPT® code 93572-26). But the equipment and FFR catheter are not currently reimbursed to the hospital per se. They are allowable, but only under what's called a Diagnosis-Related Group (DRG): a lump "bucket" sum paid to the hospital for a PCI (a.k.a. an angioplasty or stent procedure). Most ancillary equipment used, such as FFR, has to be deducted from the fixed DRG payment -- so using this advanced and recommended technology to improve outcomes actually winds up costing the hospital money -- unless, of course, the hospital takes a wider view of costs and, as Dr. Pichard discusses, realizes the savings they've gained on the decrease in complications, repeat procedures and lower use of multiple stents. But taking that wider view takes convincing. Hopefully, the boost given to Fractional Flow Reserve (FFR) by the increased level of evidence in these international guidelines will be not only convincing but also reflected in a revisited and revised reimbursement policy for tools that improve PCI outcomes. « permalink » « send comment » « back to top »
October 4, 2010 -- 8:20pm EDT IVUS for Every Stent and Angioplasty?
Read more of what Dr. Pichard says about IVUS, FFR and OCT in our exclusive interview with him , part of our Intravascular Imaging Center. « permalink » « send comment » « back to top »
September 14, 2010 -- 6:25pm EDT Training for the Transradial Approach to
Angioplasty and Stents These are major U.S. meetings, but there are other training opportunities around as well -- for example, Tejas Patel's annual TRICO meeting in Ahmedabad, India is scheduled for October 30-31. It is the meeting where a number of American interventionalists first learned the technique of angioplasty via the wrist and brought it back to the U.S. And renowned cardiologist Dr. Shigeru Saito's Kamakura Live Demonstration Course in Japan is being held in early December. If you are interested in learning the transradial approach to coronary diagnostic and treatment, visit Angioplasty.Org's page on "Transradial Training Courses" for information on upcoming training opportunities; and, if you are offering such a course, and it is not listed, please send us the information via our "Transradial Course Submission" page. The transradial wrist approach is used 50% of the time in many countries, but at last look less than 5% in the U.S. I predict that the next time these figures are published, we'll be seeing closer to 10% in the U.S. And here's why:
Yes, we're talking about a procedure for the heart...but if you look at the data, it's a no-brainer! « permalink » « send comment » « back to top »
September 13, 2010 -- 3:15pm EDT Wrist Angioplasty Training Sessions at
TCT 2010 Interventionalists have been hearing about the decreased complications, better outcomes and increased patient comfort with the wrist approach and are eager for more information. SCAI is in fact sponsoring a one-day transradial course in Boston on November 5. So make sure to register for the various TCT transradial events and get there early This year's major half-day session is on Thursday morning, from 8am-noon, in Room 145AB. Terumo Interventional Systems is also conducting one-hour workshops on Thursday, Friday and Saturday in the Training Pavilion section, which is now located inside the Exhibit Hall, near their booth #2066, and hosting a Friday evening dinner satellite symposium. Get all the details at Angioplasty.Org's Transradial Angioplasty at TCT 2010 -- we've also provided a printer-friendly version of the schedule. |
 Last
week saw the latest chapter of the year-and-a-half "Maryland
Stent Story", starring Dr. Mark G. Midei pitted against Towson-based
St. Joseph's Medical Center. Dr. Midei, accused last year by St.
Joe's of implanting almost 600 unnecessary coronary stents and
relieved of his privileges, filed a $60 million fraud lawsuit against
the hospital and its parent company. I won't go into the details
because you can read all about it in the Baltimore Sun's article
by Tricia Bishop "
Last
week saw the latest chapter of the year-and-a-half "Maryland
Stent Story", starring Dr. Mark G. Midei pitted against Towson-based
St. Joseph's Medical Center. Dr. Midei, accused last year by St.
Joe's of implanting almost 600 unnecessary coronary stents and
relieved of his privileges, filed a $60 million fraud lawsuit against
the hospital and its parent company. I won't go into the details
because you can read all about it in the Baltimore Sun's article
by Tricia Bishop "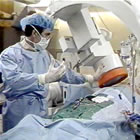 Last
month saw the publication of a joint statement on: "
Last
month saw the publication of a joint statement on: " So
only months after he had invented the procedure, he invited 28
physicians to gather in the auditorium of the hospital and watch
him perform angioplasty on seven patients via a small closed-circuit
TV monitor (pictured on right). At that point Gruentzig
had only done 27 angioplasties...period! That event, and the subsequent
live courses that he did in Zurich and then in Atlanta were the
genesis of a whole new specialty, interventional cardiology, and
a whole new type of therapy for patients -- opening blockages,
delivering heart valves, stopping heart attacks as they are
occurring(!) -- all without surgery, without opening the chest,
without using a heart-lung machine -- all done by inserting a catheter
in the groin or arm artery.
So
only months after he had invented the procedure, he invited 28
physicians to gather in the auditorium of the hospital and watch
him perform angioplasty on seven patients via a small closed-circuit
TV monitor (pictured on right). At that point Gruentzig
had only done 27 angioplasties...period! That event, and the subsequent
live courses that he did in Zurich and then in Atlanta were the
genesis of a whole new specialty, interventional cardiology, and
a whole new type of therapy for patients -- opening blockages,
delivering heart valves, stopping heart attacks as they are
occurring(!) -- all without surgery, without opening the chest,
without using a heart-lung machine -- all done by inserting a catheter
in the groin or arm artery.
 Yesterday
we posted our
Yesterday
we posted our  IVUS
has been around for more than 20 years and has been used in
only a small percentage of cath labs, although that number
is growing. So why does Dr. Augusto Pichard, Director of the
Cardiac Catheterization Lab at Washington Hospital Center,
use intravascular ultrasound (IVUS) on all of his PCI (angioplasty
and stent) procedures? He explains in
IVUS
has been around for more than 20 years and has been used in
only a small percentage of cath labs, although that number
is growing. So why does Dr. Augusto Pichard, Director of the
Cardiac Catheterization Lab at Washington Hospital Center,
use intravascular ultrasound (IVUS) on all of his PCI (angioplasty
and stent) procedures? He explains in 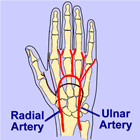 Yesterday
I wrote about the
Yesterday
I wrote about the  We
just posted a comprehensive listing of the various
We
just posted a comprehensive listing of the various