|
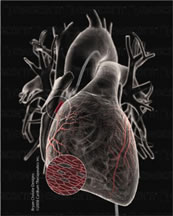
March 11, 2008 -- San Diego -- Cardium Therapeutics (Amex: CXM) announced positive findings from its preclinical study being conducted by researchers at Emory University, which demonstrated highly efficient uptake of DNA-based vectors within acutely ischemic myocardium (heart muscle lacking sufficient blood flow as a result of blockage of the coronary arteries). The study employed an occlusion and reperfusion model analogous to a heart attack or acute myocardial infarction, in which the coronary arteries supplying the heart muscle are initially blocked and later reopened, as occurs in patients after a heart attack. In that setting, delivery of an adenovector using Cardium's intracoronary administration technique resulted in robust protein expression within acutely ischemic areas of the myocardium that are most at risk for damage following a heart attack. In addition to showing high levels of uptake, there was no evidence of any increase in inflammatory responses following intracoronary infusion to the acutely ischemic heart as compared to controls.
Cardium's Corgentin (Ad5IGF-I) preclinical product candidate is a next-generation DNA-based therapeutic using the insulin-like growth factor-I gene carried by an adenovector that is being developed as a potential one-time treatment to promote the preservation and restoration of heart tissue and cardiac function following a heart attack (acute myocardial infarction). Cardium believes that Corgentin, which effectively enables prolonged IGF-I expression within the heart muscle, offers the potential to prevent further damage to heart muscle and improve cardiac healing following a heart attack. The inherent biological properties of IGF-I, which include inhibition of cardiomyocyte apoptosis, adaptive cardiomyocyte hypertrophy, recruitment of cardiac progenitor cells, as well as the induction of angiogenesis and enhancement of cardiac function, combined with Cardium's delivery approach to yield sustained localized expression, provide the rationale for the development of a therapy directed at myocardial repair and restoration. This combination of positive biological attributes and effective delivery supports the potential of Corgentin to improve functional recovery and prevent ventricular dysfunction and the associated progression to congestive heart failure following myocardial infarction and reperfusion. According to the American Heart Association, the estimated annual incidence of myocardial infarction (MI) is 600,000 new attacks and 320,000 recurrent attacks. Even with the best of care and successful early intervention, such as tissue plasminogen activator (tPA) or emergency angioplasty, about 30% of heart attack patients will eventually go on to develop decompensation through ventricular remodeling and progressive congestive heart failure. This explains in large part why heart failure remains an epidemic health problem despite improved treatments for acute cardiac events. The safety of systemic IGF-I protein therapy has been confirmed in multiple human clinical studies for a number of medical indications, and has been approved by the FDA for long-term use in children in connection with Tercica's Increlex™ product (generic name mecasermin). While considered generally safe, systemic IGF-I protein delivery is not believed to effectively target cardiomyocytes and other critical cells within the heart in a localized and sustained manner. Preclinical studies suggest that by targeting the heart using intracoronary infusion of DNA-coded, myocardial-directed delivery, using the methods pioneered for Cardium's Generx™ development program, Corgentin has the potential to induce a positive biologic response in affected heart muscle. The targeted cells are believed to produce therapeutic protein levels directly in the myocardium where it is needed. There have been an estimated 1,000 patients that have been treated with various dose levels of IGF-I protein, and now over 450 patients who have received Generx (Ad5FGF-4, alferminogene tadenovec, a DNA-based angiogenic growth factor) via intracoronary administration. We believe the safety and preliminary efficacy from these studies provide further support for the clinical potential of Corgentin. About Cardium Cardium Therapeutics, Inc. and its subsidiaries, InnerCool Therapies, Inc. and the Tissue Repair Company, are medical technology companies primarily focused on the development, manufacture and sale of innovative therapeutic products and devices for cardiovascular, ischemic and related indications. Cardium's lead product candidate, Generx™ (alferminogene tadenovec, Ad5FGF-4), is a DNA-based growth factor therapeutic being developed for potential use by interventional cardiologists as a one-time treatment to promote and stimulate the growth of collateral circulation in the hearts of patients with ischemic conditions such as recurrent angina. For more information about Cardium Therapeutics and its businesses, products and therapeutic candidates, please visit http://www.cardiumthx.com or view its 2006 Annual Report at http://www.cardiumthx.com/flash/pdf/2006CardiumAnnualReport.pdf. Cardium's InnerCool Therapies subsidiary is a San Diego-based medical technology company in the emerging field of temperature modulation therapy to rapidly and controllably cool the body in order to reduce cell death and damage following acute ischemic events such as cardiac arrest or stroke, and to potentially lessen or prevent associated injuries such as adverse neurological outcomes. For more information about Cardium's InnerCool subsidiary and therapeutic hypothermia, including InnerCool's Celsius Control System™, which has received regulatory clearance in the U.S., Europe and Australia, and its CoolBlue™ surface-based system, which is currently being marketed in the U.S., please visit http://www.innercool.com. Cardium's Tissue Repair Company subsidiary (TRC) is a San Diego-based biopharmaceutical company focused on the development of growth factor therapeutics for the treatment of severe chronic diabetic wounds. TRC's lead product candidate, Excellarate™, is a DNA-activated collagen gel for topical treatment formulated with an adenovector delivery carrier encoding human platelet-derived growth factor-BB (PDGF-BB). Excellarate™ is initially being developed to be administered once or twice for the potential treatment of non-healing diabetic foot ulcers. Other potential applications for TRC's Gene Activated Matrix™ (GAM) technology include therapeutic angiogenesis (cardiovascular ischemia, peripheral arterial disease) and orthopedic products, including hard tissue (bone) and soft tissue (ligament, tendon, cartilage) repair. For more information about Cardium's Tissue Repair Company subsidiary, please visit http://www.t-r-co.com. Forward-Looking Statements Except for statements of historical fact, the matters discussed in this press release are forward looking and reflect numerous assumptions and involve a variety of risks and uncertainties, many of which are beyond our control and may cause actual results to differ materially from stated expectations. For example, there can be no assurance that results or trends observed in preclinical studies will be reproduced in subsequent studies, that human clinical trials can be conducted and completed in an efficient and successful manner, that product modifications or launches will be successful or that the resulting products will be favorably received in the marketplace, that our products or proposed products will prove to be sufficiently safe and effective, that necessary regulatory approvals will be obtained, or that our products or product candidates will not be unfavorably compared to competitive products that may be regarded as safer, more effective, easier to use or less expensive. Actual results may also differ substantially from those described in or contemplated by this press release due to risks and uncertainties that exist in our operations and business environment, including, without limitation, risks and uncertainties that are inherent in the development of complex biologics and the conduct of human clinical trials, including the timing, costs and outcomes of such trials, and our ability to obtain necessary regulatory approvals and expected qualifications, our limited experience in the development, testing and marketing of therapeutic hypothermia devices and whether our efforts to accelerate the commercialization of such devices and launch new devices will be successful or completed within the timeframes contemplated, our dependence upon proprietary technology, our history of operating losses and accumulated deficits, our reliance on collaborative relationships and critical personnel, and current and future competition, as well as other risks described from time to time in filings we make with the Securities and Exchange Commission. We undertake no obligation to release publicly the results of any revisions to these forward-looking statements to reflect events or circumstances arising after the date hereof. Cardium Therapeutics™ and Generx™ are trademarks of Cardium Therapeutics, Inc. Tissue Repair™, Gene Activated Matrix™, GAM™ and Excellarate™ are trademarks of the Tissue Repair Company. InnerCool Therapies(R), InnerCool(R), Celsius Control System™, Accutrol™, CoolBlue™ and RapidBlue™ are trademarks of InnerCool Therapies, Inc. Source: Cardium Therapeutics, Inc. |