 Sorry Jar-Jar. We all know that Episodes 1-3 of “Star Wars” just didn’t cut it when compared with the original trilogy. Although these prequels were technically more sophisticated, and the digital rendering was way more advanced, the stories were just…boring.
Sorry Jar-Jar. We all know that Episodes 1-3 of “Star Wars” just didn’t cut it when compared with the original trilogy. Although these prequels were technically more sophisticated, and the digital rendering was way more advanced, the stories were just…boring.
I started the wordplay about “Stent Wars” over a decade ago, a time when the competitive juices of the two device-makers, Boston Scientific and J&J/Cordis, were at an all time peak — each company was heavily marketing their first generation drug-eluting stent and every interventional cardiology meeting was saturated with “Cypher vs. Taxus” messaging.
I still remember walking past the J&J/Cordis expo booth at a TCT just as a late-breaking trial showing Cypher besting Taxus was announced on the video monitor feed. The sales force erupted in jump up-and-down football cheers. Go go go Cypher! Who were the Rebels and who was the Empire? Didn’t matter. It all depended upon which side you were aligned with. The “other” side was the enemy. Good times. Stent Wars.
But, exciting as that was, the whole marketing message of “our stent beats your stent” has just become so Early 21st Century. Times have changed: Cordis is no longer in the coronary stent business and Boston Scientific is trading at 15% of what it was back then. And devices have changed: they have all gotten much better. These days, most drug-eluting stents studies show outcomes that are pretty close. For example, see our recent report on the two-year TWENTE outcomes, comparing Medtronic’s Resolute and Abbott’s Xience. Spoiler alert: they’re pretty much the same! Clinical trials are now designed to show “non-inferiority,” and advances for the most part are taking the form of expanded indications that are backed up by extensive datasets. (Examples are Medtronic’s diabetes indication for its Resolute Integrity and Boston Scientific’s AMI indication for its ION and Liberte.)
Technically these are more sophisticated devices, and maybe a bit more boring but, of course, all very good news for patients….
So I recently was taken back in time, back to those “Stent Wars” days, just a tad when I got a company “e-blast” from Boston Scientific, touting the superiority of its latest stent. The email was titled “PROMUS Element Beats Xience — View the Data.”
Hmmm. I hadn’t heard this breaking news. So I did as directed. I wanted to “view the data,” so I clicked on the link in my email…and this is what I saw!
Wow! Actual explosions. Pow! Wham! And vintage 1984 techno-corp electro-pop music. And fast-moving animations of words, numbers and graphs. Very fast. So fast I couldn’t read them too closely. And that, I believe, was the point.
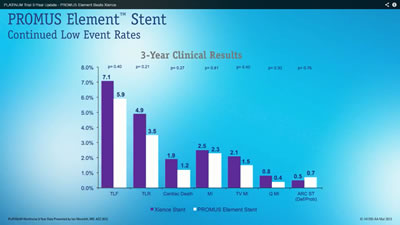
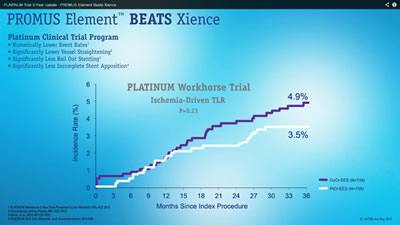
So I screen-grabbed the two main graphs in the presentation and quickly discovered why I hadn’t heard about this breakthrough study. I invite you also to “View the Data” below. The white PROMUS Element shows lower event rates than the purple Xience, so it must be better, right? Well, just click on each graphic for the full hi-res image and read on:
The important data to view? The p-values, of course. And none of them showed statistical significance. They ranged from 0.21 to 0.80; statistical significance kicks in at around 0.05, or 0.01, depending on the pre-determined cut points. For example, the Target Lesion Revascularization (TLR) graph above shows a 0.21 p-value, meaning that there’s a 21 in 100 possibility that this result occurred entirely by chance. Yes, there’s a numeric difference in the outcome, but this is not considered statistically significant.
So, music and fast-moving animation aside (did I mention very fast-moving animation?) the good old days of “Stent Wars” just don’t exist here anymore. There’s no statistical significant difference in the Xience V or PROMUS Element stents for major clinical outcomes at three years, just as recent analyses have shown no significant difference between Xience V and Resolute.
Are there any differences? You’d have to ask the operators in the cath labs. Some interventionalists have preferences; some find certain stents easier to insert than others; some hospital systems find better price value with one stent over another. Oddly enough, one of the interesting aspects of the Boston stent is its Platinum metal platform, which contains less nickel than other stents, something that may be of interest to patients who suffer from severe nickel allergy. But nowhere in the above presentation is that mentioned.
The reason I’ve gone on about this probably more than I should is that I get bothered when scientific data is cherry-picked and manipulated to promote one product over another when no real differences exist. Especially when some of the real issues in coronary devices today are about appropriate use, targeting the right patients, patients understanding what these devices can do and how to help them do that, organizing systems to provide emergency angioplasty treatment of heart attack for patients, saving lives and, you know, that sort of boring stuff — kind of like Star Wars: Episodes 1-3….




I’m looking at this information, trying to find actual DATA on results of DAPT stopped as soon as possible.
There are very few papers (I’ve found two so far) on the results of quitting DAPT after a month or three. I think it’s because no one wants to do the studies – lest the drug companies jump on them. Two 2013 Medtronics papers on their RESOLUTE stent used a pool of patients who stopped using Plavix for at least 14 days for various reasons after the first month – and found ZERO adverse effects in 1700+ patients. This pool of people to study must exist for every stent and drug protocol out there – but the studies are not being done.
I think it’s my right to make an informed decision that minimizes use of something – the consents I’ve ‘signed’ were based on cardiologist ‘recommendations’ based on guidelines – the primary data is hidden. I feel bullied into them.
Other people may be okay with using recommendations based on NO science; I am not. I find most papers lie, exaggerate their results, misuse statistics very badly, have drug company support, ignore studies which don’t support their pre-conceived notions… It’s just as bad on the stents and the DAPT as it is in the statin papers.
Thanks for saying “I get bothered when scientific data is cherry-picked and manipulated to promote one product over another when no real differences exist.” I’m looking forward to reading a lot more of your posts.
Alicia – Thanks (again) for your comments. The duration of DAPT is a major major topic at every cardiology meeting. This is not something that’s dismissed by the profession, because the downside of long duration DAPT can be bleeding complications; the downside of too short duration can be stent thrombosis. Each case is different and the “risk” involved has many components, for example, length of stent, location of stent, brand of stent, clinical picture of the patient, and so on. There are scoring protocols to help determine the ideal length of DAPT, and when it is safe to stop. So it’s definitely NOT a one-size-fits-all situation.
Thanks for your replies, and I’m looking forward to hearing about the recent meeting, especially to see if there are any changes in the recommendations.
Additional downside of DAPT is continuing side effects, and such ‘minor’ details as liver damage. Need to take NSAIDs and other pain medications is a reality for many patients. And may increase the chance of bleeding, which is already a problem.
I find it very hard to get through to my cardiologists that my Chronic Fatigue Syndrome – and the Celebrex which is the only pain med to every work for me in 27 years – are significant features inme as a patient, and need to be considered seriously. I don’t tolerate most medicines, possibly because my liver seems to take forever to clear them (based on how long a small amount of alcohol keeps me ‘drunk’).
They say ‘1 year’ or more. Period. I don’t want to go past a month for Effient, if not strictly necessary. I’m at the 1-month mark right now, and looking for data. Supposedly it only takes a month to epithelialize the inside of the stents. I found one paper on the cohort of stent possessors who stopped taking their DAPT after a month-a year for various reasons; the data for this cohort must exist for almost every other study – but I don’t see anyone anylyzing it. The people who stopped, by the way, had no problems.
Not trying to be difficult; just want important decisions to be based on actual evidence, not guesstimates and hype.